|
Whilst sulfuric acid (H 2SO 4) ionises to form sulfate ions (SO 4 2-), you can’t ionise sulfate ions any further. Water does ionise in solution, forming H + and OH – ions, but as you’ll discover post-GCSE, its ionisation is negligible compared to other ionic substances. You should also not ionise water in ionic equations. When that happens, don’t ionise the precipitate that has the state symbol (s) – it is formed as a precipitate because it’s not soluble and so won’t break up to form ions. This is especially important for precipitation reactions, where solutions are mixed and an insoluble solid forms. Lastly, don’t ionise solids or gases present as reactants or products in chemical reactions, and don’t ionise liquids unless it’s a molten ionic compound. Insoluble, except sodium, potassium, barium and ammoniumĪnything that this table indicates is soluble can be ionised in a chemical reaction. Insoluble, except sodium, potassium and ammonium If the chemical equation doesn’t include state symbols, or if it’s an aqueous substance but you’re not 100% certain that it’s soluble, you can use the solubility rules:Ĭommon salts of sodium, potassium and ammonium (Note that Cl 2 in water does form ions, but it’s through reaction with water and not what we’re considering here). The exception to this is when (aq) means a substance has dissolved without ionising.Īn example of this would be Cl 2(aq), or chlorine water, which you wouldn’t show ionising to form Cl + and Cl – for instance. 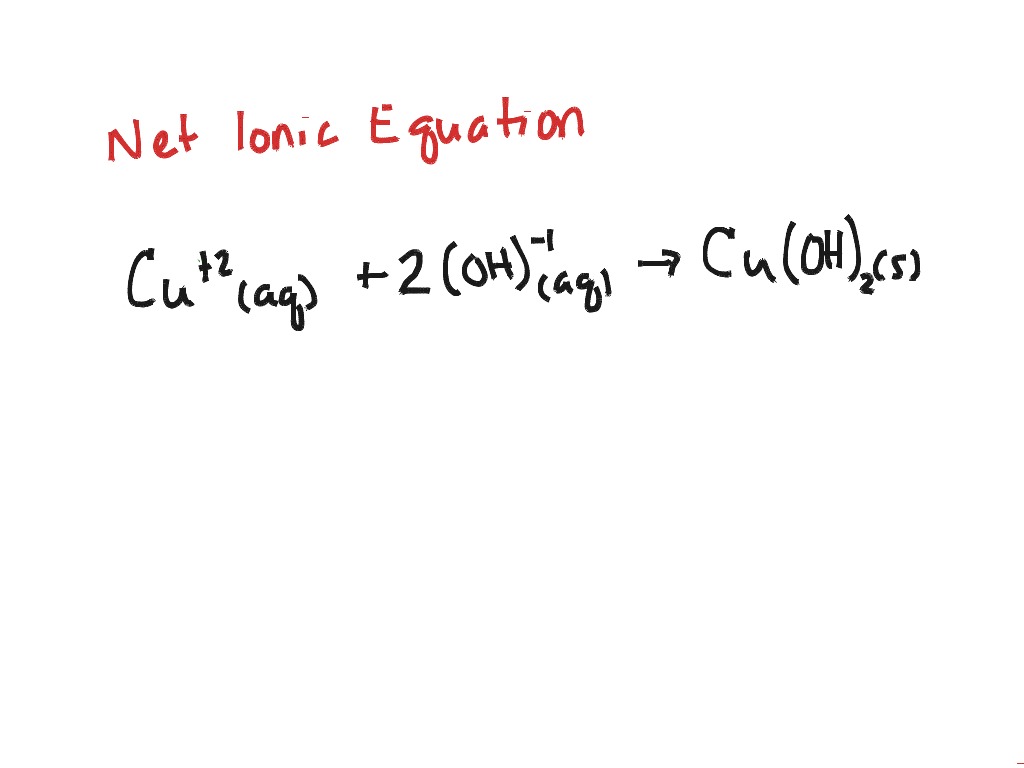
If a substance has the state symbol aqueous in a chemical equation, it means that the substance is soluble and will usually be ionised. With very few exceptions, a substance in aqueous solution will be ionised, because the process of dissolving usually requires ionisation. This we show using the state symbol aq, meaning the substance has formed an aqueous solution. 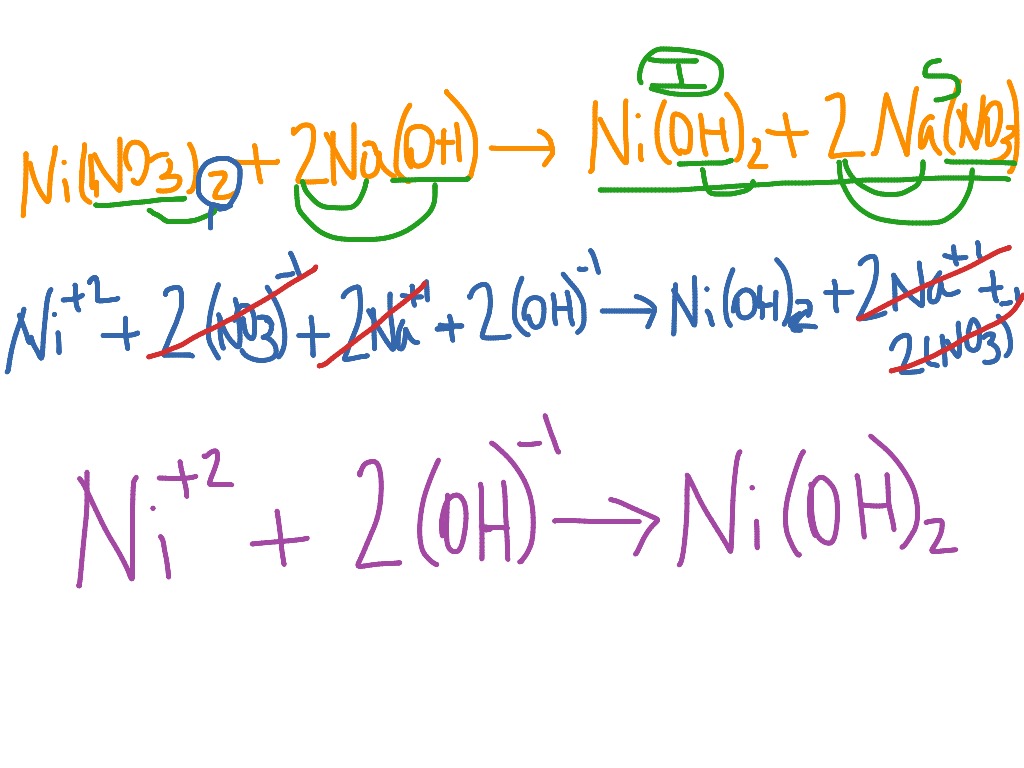
We can write the balanced equation for this:Ī substance being split up by the action of water means it has formed a solution in water – it has dissolved. In the neutralisation reaction between dilute hydrochloric acid and sodium hydroxide solution, a salt and water are formed. This is very useful, particularly when dealing with redox reactions which are complicated enough without spectator ions getting in the way. 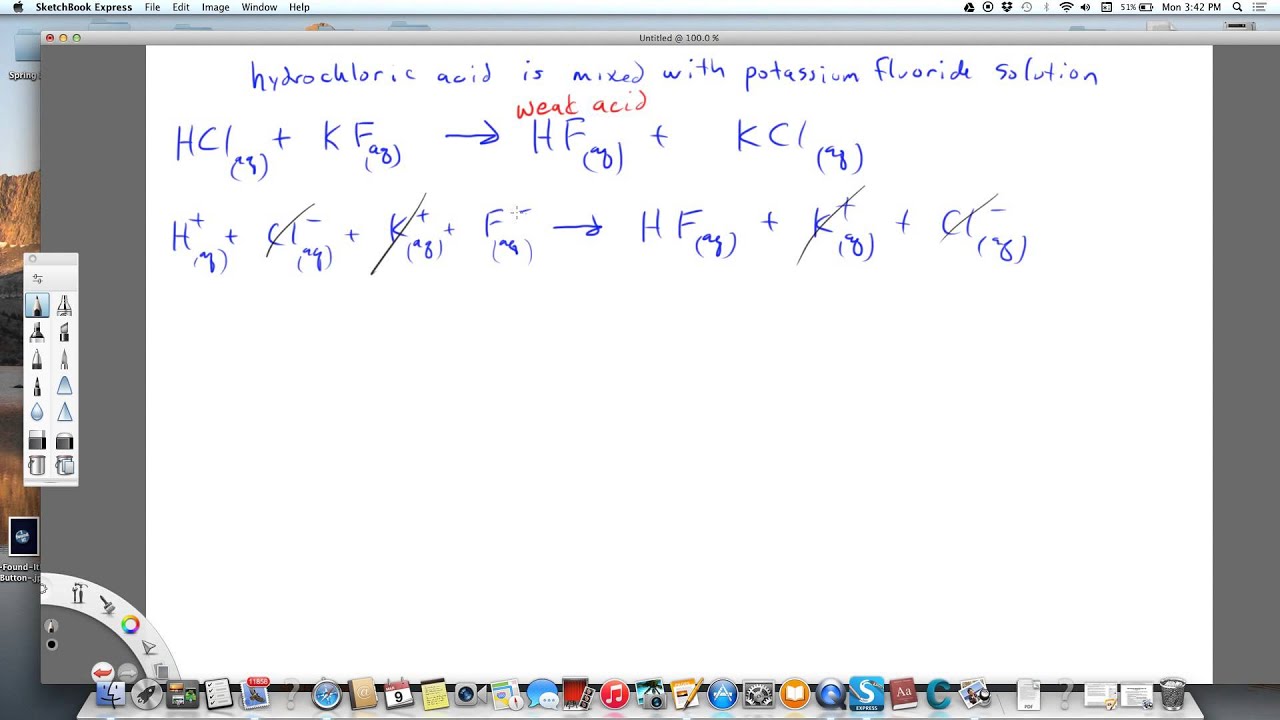
The purpose of ionic equations is they simplify chemical equations by eliminating the species that aren’t undergoing any change. The other ions that don’t participate are called spectator ions. In other words, the ions that react together in solution and form new substances. Ionic equations are chemical equations that show only ions that participate in a chemical reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
